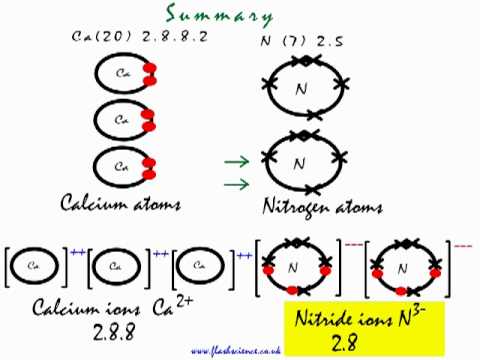
It decomposes to form carbon dioxide when heated up to 1200K. PCC has smaller particles that have higher purity, are less abrasive, and tend to have higher brightness than GCC. 
The shape of the PCC crystal depends on the product, and the particles are more regular and uniform with a distribution of narrow size. Precipitated Calcium Carbonate - This is produced via chemical precipitation with a carbocation process or as a by-product of a few bulk chemical processes. The GCC crystal shape is irregularly rhombohedral, and its distribution is a broader size. Ground Calcium Carbonate - It is produced via extraction and processing naturally occurring deposits. Both these grades compete industrially primarily based on the particle size and the characteristics that are imparted to a product. However, if there is an excess passing of carbon dioxide, it results in the formation of soluble calcium hydrogen-carbonate.Ĭommercial Production of Calcium CarbonateĬalcium carbonate can be produced commercially in two different grades. It can be prepared on a large scale by passing carbon dioxide gas through calcium hydroxide (otherwise called slaked lime). When carbon dioxide is passed in excess amounts, it leads to the formation of calcium hydrogen-carbonate. There is also another method to obtain the calcite by adding sodium carbonate to calcium chloride. When carbon dioxide compound is passed through slaked lime, calcite is obtained. We can find it in extensive usage in the manufacturing industry as an ingredient for quick lime and cement and building material (marble).ĬaCO 3 is obtained mainly by using slaked lime and carbon dioxide as raw materials. It is added to swimming pools as a disinfectant agent and also as a pH corrector. It can also be used as cosmetics fillers. Medicinally, we use it as an antacid or a calcium supplement. It is a white and insoluble powder-like substance that occurs naturally in minerals, marble, chalk, limestone, shells, calcite, pearl, and other related compounds. The Calcium Carbonate structure can be given as follows:Ĭalcium Carbonate is a chemical compound having the chemical formula CaCO 3. CaCO 3 StructureĬalcium carbonate is odorless, and a non-toxic compound found commonly as a white mineral that occurs naturally in limestones, chalks, and marbles. It finds its uses in regenerative medicine and a range of personal care products. It is found naturally in springs, organic tissue, urinary calculi, gallstones, and plants. Vaterite: Hexagonal in shape, vaterite, is less stable than calcite and aragonite, with a higher solubility. It is used to remove pollutants like zinc, cobalt and lead from contaminated wastewaters. Aragonite provides ingredients to sea life, and also maintains the pH levels to its natural level. It is formed by biological and physical processes, including precipitation from marine and freshwater environments. 
It is used extensively in construction material, agricultural soil treatment, pharmaceuticals and many more areas.Īrgonite: It is less stable and more soluble than calcite. Calcite is a common constituent of sedimentary rocks like limestone and marble. A rock-forming mineral, it is commonly found in sedimentary, metamorphic, and igneous rocks. Types of Calcium Carbonate found in the EnvironmentĬalcium carbonate is present in three polymorphic forms - calcite, aragonite and vaterite.Ĭalcite: It is the most stable and the least soluble. The symbol of calcium carbonate can be given as CaCO 3. Although they are available in different forms, they are chemically similar and differ only physically. It is available in various forms, such as limestone, marble, and more. It is also one of the most popular chemicals which is encountered first in school classrooms, where the use of chalk (which is a form of CaCO 3 ) is found. However, Star Wars is science fiction and may have even been the reason for the choice of using the word carbonite.Calcium carbonate is a chemical inorganic compound having the chemical formula CaCO 3. Some of you reading this might be saying right now, "Wait a minute! I remember hearing about carbonite in Star Wars!" That's true. Even though the carbonate ion, CO 3 2 –, is quite common, there is no such thing as the carbonite ion. It should also be pointed out that just because the -ite form always has one less oxygen than the -ate form doesn't mean that an -ite form is possible. Furthermore, if you learn the one that ends with -ate, the ion that ends with -ite can easily be obtained, since it always has one less oxygen than the -ate form. It is also the reason why memorizing the form that ends with -ate is recommended rather than the one that ends with -ite. This is because the -ate form is more commonly found in nature than the -ite form.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
